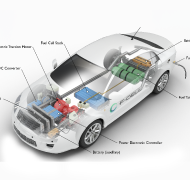
Fuel Cell Electric Vehicles
Fuel cell electric vehicles (FCEVs) are powered by hydrogen. They are more efficient than conventional internal combustion engine vehicles and produce no harmful tailpipe pollutants—they only emit water vapor and warm air. FCEVs and the hydrogen infrastructure to fuel them are in the early stages of implementation. The U.S. Department of Energy leads research efforts to make hydrogen-powered vehicles an affordable, environmentally friendly, and safe transportation option. Hydrogen is considered an alternative fuel under the Energy Policy Act of 1992 and qualifies for alternative fuel vehicle tax credits.
What is a fuel cell electric vehicle?
FCEVs use a propulsion system similar to that of electric vehicles, where energy stored as hydrogen is converted to electricity by the fuel cell. Unlike conventional internal combustion engine vehicles, these vehicles produce no harmful tailpipe pollutants. Other benefits include increasing U.S. energy security through diversity of energy sources and strengthening the economy.
FCEVs are fueled with pure hydrogen gas stored in a tank on the vehicle. Similar to conventional internal combustion engine vehicles, they can fuel in about 5 minutes and have a driving range of more than 300 miles. FCEVs are equipped with other advanced technologies to increase efficiency, such as regenerative braking systems that capture the energy lost during braking and store it in a battery. Major automobile manufacturers are offering a limited but growing number of production FCEVs to the public in certain markets, in sync with developing supporting infrastructure.
How Fuel Cells Work
The most common type of fuel cell for vehicle applications is the polymer electrolyte membrane (PEM) fuel cell. In a PEM fuel cell, an electrolyte membrane is sandwiched between a positive electrode (cathode) and a negative electrode (anode). Hydrogen is introduced to the anode, and oxygen (from air) is introduced to the cathode. The hydrogen molecules break apart into protons and electrons due to an electrochemical reaction aided by a catalyst. Protons then travel through the membrane to the cathode.
The electrons are forced to travel through an external circuit to perform work (providing power to the electric motor) then recombine with the protons on the cathode side where the protons, electrons, and oxygen molecules combine to form water. See the Fuel Cell Electric Vehicle (FCEV) infographic to learn more about the process.